- Adobe illustrator cs3 classroom
- Cross dj free review
- Reflector 2 license key generator
- Uninstall msi burn recovery
- Chemical equation balancer calc
- Starbound change cursor color
- Steinberg hypersonic 2 rar
- The tudors season 1 episode 1 who sleeps with daughter
- Consider a carnot cycle heat pump
- Sonik synth 2 ye
- The 22 immutable laws of marketing torrent
- True pokemon emerald randomizer
- Gaffer sinhala cartoon
- Empire earth 4 download free
- Is there a working ps3 emulator for pc
- Lost pause plays tokyo school life
- Bootstrap templates responsive emp
- Ice xp sound of music halo
- Cara membuat program kasir
- The incredible hulk 2008

Therefore, the efficiency of the Carnot heat engine is: We can simplify this expression by using the equation of an adiabatic process for processes 2-3 and 4-1:Īnd after dividing both expressions, we obtain the following relation between the volumes: In the Carnot cycle they correspond respectively to the heat absorbed in process 1-2 and to the heat released in process 3-4, because the two other processes are adiabatic and therefore occur without transferring heat.Īfter substituting the values of Q 1 and Q 2 in the efficiency expression we have:
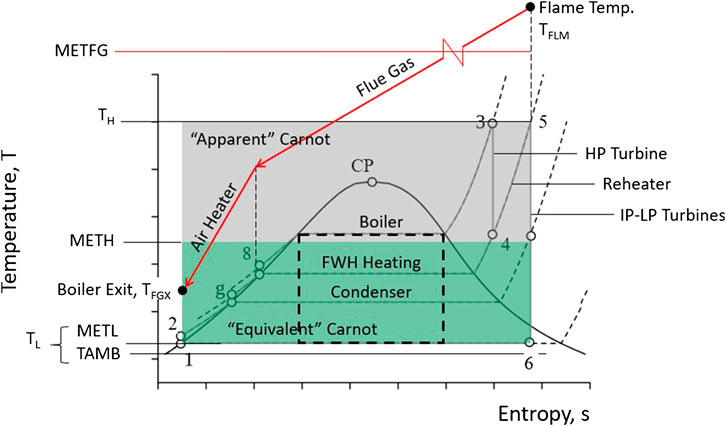
Where Q 1 is the heat absorbed by the working fluid and Q 2 is the heat discharged to the hot reservoir. The efficiency of a heat engine is given by: The Carnot cycle is illustrated on a PV diagram in the following figure:Īs you can see in the figure above, the cycle goes clockwise, because a heat engine does work on its surroundings (W>0). 4-1 Adiabatic Compression: to close the cycle, the ideal gas is compressed adiabatically and its temperature rises back to the temperature of the hot thermal reservoir.During this process, it discharges an amount Q 2 of heat energy to the cold thermal reservoir. 3-4 Isothermal Compression: the ideal gas is compressed isothermally in contact with the cold thermal reservoir.2-3 Adiabatic Expansion: the ideal gas expands adiabatically, it cools to the temperature T 2 of the cold thermal reservoir.During this process the gas absorbs a quantity Q 1 of heat from the hot thermal reservoir. 1-2 Isothermal Expansion: the ideal gas is allowed to expand at constant temperature because it is in contact with the hot thermal reservoir at temperature T 1.The Carnot cycle consists of four reversible processes: There are three types of heating pumps in total: air, ground or water source.A Carnot heat engine is an ideal heat engine that, as we will show below, has the maximum efficiency that a heat engine can achieve when operating between two thermal reservoirs.įirst, we will determine the performance of this heat engine assuming that its working fluid is an ideal gas. It is even known that this savings is over 50%. Compared to fossil fuels, heat pumps provide much higher savings. Heating pumps are preferred for heating in winter and cooling in summer without any problem. As a result, the transfer to the distribution system takes place.
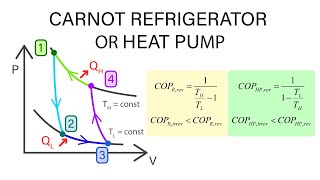
Then, high temperature is provided with the steam transferred to the compressor. The low-temperature heat taken from the air, water or soil is somehow converted into steam thanks to the refrigerants. This type of pumps can be used alone or with an additional system. In fact, the working principle of heating pumps is technically no different from a simple refrigerator that we use in our homes today. Heating pumps efficiently use the low-temperature heat they obtain from nature. The energy crisis that emerged in the global world in 1973 makes heating pumps popular again. Although important developments have been observed in these pumps after the 1950s, heat pumps begin to fall out of favor due to the relatively high cost of establishment. In the period just before World War II, many engineers worked intensively on the development of heating pumps. In this way, the heating pumps are activated for the first time. Lord Kelvin suggests that refrigeration machines can actually be used for heating purposes. Essentially known as a type of refrigeration cycle, heating pumps underwent a serious transformation in 1850.

Nicolas Carnot introduced the basic principles of the heat pump in 1824. What is the Working Principle of Central Heating Pump?